A Food and Drug Administration advisory panel Friday afternoon voted against recommending the approval of booster doses of a COVID-19 vaccine for the general public, saying it needed more data. But the group unanimously approved a second, narrower recommendation for booster shots for older adults and individuals at high risk for severe disease, including healthcare workers.
The Vaccines and Related Biological Products Advisory Committee met Friday to review an emergency use authorization application from Pfizer and BionNTech for a third dose of its COVID-19 vaccine. The application sought to administer a booster dose at least six months after the second shot for people aged 16 or more years.
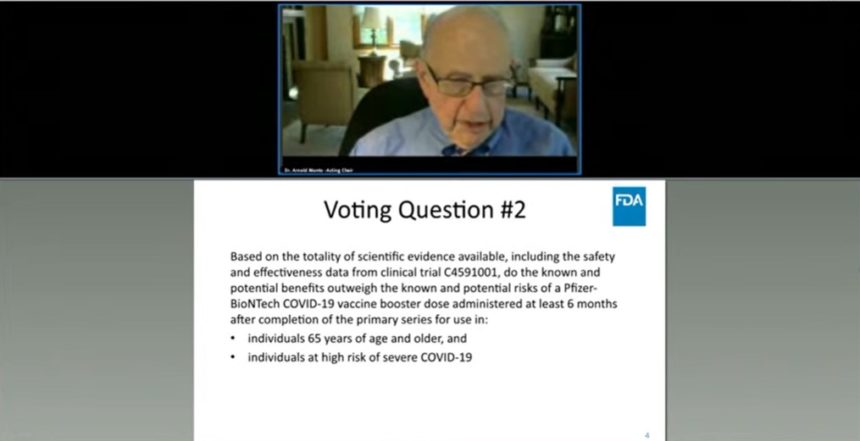
Although the advisory panel voted 16-2 against approving a booster shot for the general population, the group unanimously approved a third dose for those 65 and older and for individuals at high risk of severe disease. A poll of members showed unanimous support for including “healthcare workers or others at high risk for occupational exposure” in the emergency use authorization for a booster dose.
The FDA does not have to accept the VRBPAC panel’s recommendations and still must weigh in on authorizing booster doses of the Pfizer vaccine. If the FDA approves the boosters, the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices, scheduled to meet Wednesday and Thursday, then will refine the recommendation on who should receive the shots.
The FDA released briefing documents ahead of Friday’s meeting indicating that the current vaccine still adequately protects against the virus. A CDC study released Friday found that the Moderna vaccine was more effective than the Pfizer and Johnson & Johnson vaccines several months after vaccination but that all three were still highly effective at preventing hospitalization.
The Biden administration announced plans last month to begin rolling out booster shots to all eligible Americans beginning next week, subject to FDA review and CDC recommendations.




