As the senior living industry awaits today’s scheduled vote by the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices on how to implement COVID-19 booster shots for older adults and healthcare workers, providers are working behind the scenes to prepare for on-site clinics.
Bri Morris, director of strategic initiatives at National Community Pharmacists Association, discussed connecting providers with local pharmacies for onsite booster clinics, as well as ways to address resident and staff vaccination, during a LeadingAge membership call on Wednesday.
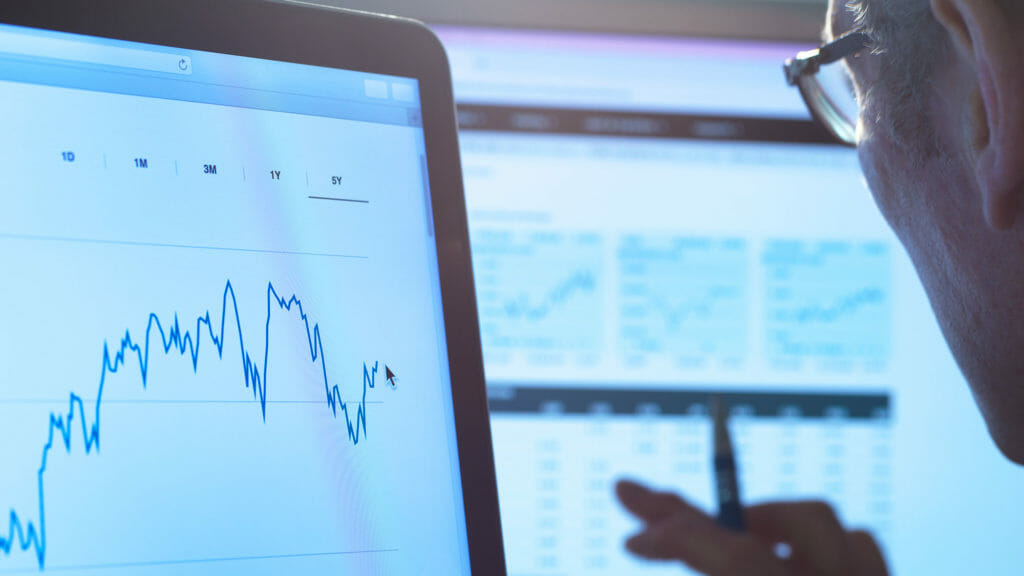
Morris said that as of Sept. 8, almost 127 million COVID-19 vaccine doses have been given through pharmacies. Her organization now is working with community pharmacies to contact long-term care facility partners to set up booster clinics and establish best practices, she added.
While everyone is waiting for information about the plan to deliver booster shots, Morris said, annual flu shots can be given at the same time as COVID-19 boosters, providing a way for both providers and pharmacies to maximize their time.
Tuesday, the CDC rolled out new vaccine access resources for long-term care settings, including assisted living communities, residential care communities, group homes and other forms of senior housing. The resources include strategies for on-site vaccination clinics and vaccinations in the broader community. The CDC also prepared a booster shot page that will be updated if and when booster doses are authorized.
Providers also can request vaccination support from a pharmacy partner in the Federal Retail Pharmacy Program or find information about connecting with federal pharmacy partners on the CDC website.
The Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee last week unanimously approved the emergency use authorization application from Pfizer–BioNTech for a third dose of its COVID-19 vaccine for older adults and individuals at high risk for severe disease, including healthcare workers. The group voted against recommending the booster doses for the general public.
Last night, the FDA amended its emergency use authorization for the Pfizer–BioNTech vaccine, now marketed as Comirnaty, to allow for use of a single booster dose to be administered six months after the last dose, for those 65 and older, those aged 18 to 64 “whose frequent institutional or occupational exposure to SARS-CoV-2 puts them at high risk of serious complications of COVID-19 including severe COVID-19,” and for others aged 18 to 64 at high risk of severe COVID-19.
The CDC’s advisory panel, which met yesterday and will wrap up discussions today, will outline more precise recommendations for how booster shots will be administered.